Neotropics

Turrialba Volcano
and the Infrastructure of Everything
On Costa Rica's Turrialba Volcano, a journey into the living systems beneath the world. Part 1 of 3.
Updated March 2, 2026
Iam standing on a sharp bend in the road on Turrialba Volcano with naturalist guide Harry Barnard and Fernanda, a high school student pursuing her proyecto de investigación in ecotourism.
At this elevation, we are surrounded by undisturbed native cloud forest, dense with life. Lobsterclaws hang in the understory. Pink bananas, massive philodendrons, and monsteras rise in a tangled wall of green, while living-vase bromeliads grow from towering canopy trees. Below us, the land falls away into cloud and forest, rivers beginning journeys they will not complete for hundreds of miles. Everything here is connected to something it cannot see.
In Costa Rica, seniors in high school are required to complete a proyecto de investigación, an original research project, before graduation. It’s an unusual expectation, but it’s very Costa Rican in its expectation that young people are capable of producing knowledge, not just absorbing it. Fernanda’s project focuses on ecotourism, but for her this world shrouded in a smoking volcano above is not theoretical. She grew up here on Turrialba as the daughter of a farming family, watching weather patterns shift, crops struggle, and forests retreat or recover. She was aware of the birds and wildlife of the land she grew up with, but you cannot really start to see the diversity until you learn it with somebody like Harry.
For me, as someone who grew up always poking around in the woods, I realize Fernanda is seeing her backyard in a way that I could not comprehend at age eighteen. I never imagined that in the very woods in Minnesota I haunted every summer day, I could see colorful Summer Tanagers and Baltimore Orioles, Jack-in-the-Pulpits, Ghost Pipes and Red Admirals. But today, Fernanda will see her backyard in a way that will likely alter the way she sees it forever.
The life is overwhelming. Costa Rica’s Cartago Province is not well known on the travel circuits, yet it is a haven of biodiversity. I am staying at a small lodge, Rancho Naturalista, which sits at a rare crossroads between the Pacific and the Caribbean, as well as the lowland and the highland. The 120-acre property lies at a perfect intersection of natural zones, and because of that it holds the country’s highest known bird diversity, with 527 species recorded on the grounds. Even Pacific and Caribbean seabirds have been seen drifting overhead.
Costa Rica itself occupies a kind of fairytale zone of biodiversity. Species richness peaks in the tropics. A majority of life on Earth exists in the tropics, a pattern known as the latitudinal diversity gradient. Life concentrates most densely in the warm, wet band around the equator and thins steadily toward the poles.
As we scan a mixed flock of high-elevation birds, I think of an article by Jon Miltimore that I debunked months ago. It relied on an infographic that tried to reduce biodiversity to the fate of a single Arctic species, a place where diversity is naturally sparse. The piece was riddled with errors and misused citations, but standing here, that almost feels beside the point. The deeper mistake was conceptual. It assumed that the complexity unfolding in front of me could be collapsed into a single symbol.

A spiny-headed treefrog rests in bamboo, where she protects her tadpoles.
The distortion was the idea that one charismatic animal could stand in for the condition of an entire living planet. A polar bear becomes a proxy for Earth. Win the bear and the argument is settled. Lose the bear and everything is supposedly fine. The appeal of this framing lies in how easily it simplifies a world whose underlying systems are anything but simple.
What keeps bothering me on Turrialba is that even highly educated people, people who read science, who accept climate change, who consider themselves environmentally literate, often do not understand what biodiversity actually means for human civilization. It’s not their fault; there is almost no one attempting to explain biodiversity at the level where it becomes legible. It is usually presented as facts, species counts, or moral obligation. It is easier to say that Monarch butterflies are in decline than to describe something more web-like, more labyrinthine, more complex.
What is rarely articulated is that biodiversity is the condition that makes a habitable world possible in the first place.
The thought I cannot shake is a simple one. Biodiversity is a structural fact. Human societies do not sit first on technology or ideology or economics. They sit on living systems. They are built on the biosphere. Understanding that, and acting in its preservation, is humanity’s most urgent task.
That claim can sound abstract until it is placed alongside real human urgency. If your child has cancer, then curing cancer becomes everything. If you are in Ukraine and Russian bombs are killing your neighbors and stripping away your future, then the death of Putin is the only thing that matters.
None of this is theoretical. These struggles are real, immediate, and not interchangeable. Yet each one assumes something that rarely gets named. They presume a world that still functions underneath them. Medicine, research and human rights depend on societies that still hold together. Even war does not occur in isolation. Its causes, its scale, and what follows are shaped by shortages, environmental stress, and the slow breakdown of systems people rely on.
This is not an argument against caring deeply about human suffering. It is an argument about ordering reality correctly. Solving for cancer, defending human rights, and resisting authoritarian violence are urgent human imperatives. They all sit downstream of biodiversity. Without living systems that grow food, regulate climate, clean water, and absorb shocks, moral victories do not endure. Rights, laws, and institutions depend on biological stability that rarely receives attention.
That urgency is easy to miss because attention is constantly being redirected. Each year brings a new existential panic. This year, it’s artificial intelligence. Last year was social media. The year before that, culture wars. Remember Y2K?
When the noise falls away, very few forces threaten human civilization at a planetary scale. Nuclear war does. A large comet impact does. Both would be abrupt and unmistakable. Both remain statistically unlikely.

Deep Blue-eyed Metalmark, an example of the extraordinary biodiversity of Costa Rica's forests.
Biodiversity collapse belongs in this same narrow category of existential risk, with one crucial difference. It is already unfolding. Unevenly. Quietly. Largely unmanaged. There is no explosion. No singular moment.
That is why biodiversity loss is so often underestimated. It looks like normalcy slowly losing its guarantees. Crops still grow until they do not. Freshwater still flows until it becomes erratic. Ecosystems continue functioning until they suddenly fail.
A mixed flock is moving through the trees above. Flame-colored Tanagers, a Slaty Flowerpiercer, Black-thighed Grosbeaks, and a Spangle-cheeked Tanager. Harry grew up in England in a family of avid birders, and so an almost Victorian naturalist vocabulary is second nature to him. So precise is his language that small details come into focus for anyone standing alongside him; that language adds a layer of beauty to everything we observe. He’ll say things like, “there’s just a faint hint of viridescence along the trailing edge of the tertiaries,” or, “that breast isn’t brick-red, it’s closer to crushed cochineal.”
When we stand still over a roadside ledge, looking down into a dark quebrada, clouds and mist descend on us, making everything around us invisible except for the dark riverine corridor below. Harry remarks on his disdain for the name of the secretive skulking bird we are looking for. Of the Wrenthrush, he explains. “Everything about its name is unfortunate. It’s not a wren. It’s not a thrush.” He comments on its beautiful song, its bright orange crest. “It’s the only species in its genus, Zeledonia coronata, and it used to be called just that. Zeledonia. That was such a more lovely name. To me, it is still the Zeledonia.”
After we see a tiny Volcano Hummingbird resting on a fence, Harry points to a nearby plant. “This is Cavendishia, an ericad,” he says, “You see how the corollas carry that lacquered carmine, which fades into this sublime smoke-violet at the tips. That’s classic plant signaling to a hummingbird, precisely evolved for bills like Selasphorus.”
From our perch up here on Turrialba Volcano, the fact that we are so close to both the Caribbean and Pacific becomes visible. To the east, we can see the blue of the Caribbean. A few container ships are just barely visible beyond the haze. And to the west, we can sense the marine layer just beyond that Pacific slope.
Two Oceans Below
Being caught between two oceans is a reminder that some organisms, even some individual species, have global impact. Get rid of just that one species, and the world unravels.
These two oceans are where organisms exist that are doing work most people will never see. Coccolithophores are single-celled plankton that sit near the base of marine food webs. By turning sunlight and carbon into living matter, they underpin the biology of the oceans, as well as supporting the fisheries that literally feed millions.
When coccolithophores die, their shells sink. Over time, they carry carbon into the deep ocean, making them one of the world’s key regulators of carbon. They also play a role in the planet’s albedo; they reflect heat in a way that makes them regulators of climate. Over much longer stretches of time, their remains accumulate into chalk and limestone, the raw material of cliffs, buildings, and cities. A surprising amount of the physical world is built from organisms almost no one knows exist.
But the system under which coccolithophores evolved over hundreds of millions of years is changing. As carbon accumulates in the atmosphere, more of it dissolves into seawater. Ocean chemistry is changing; acidifying. Conditions these plankton adapted to over millions of years become harder to maintain. Their shells are thinning and their growth is slowing. When organisms this foundational begin to falter, the effects do not stay confined to the sea. They move upward through food webs and outward into climate regulation, narrowing the margin of safety complex societies depend on. The effects accumulate slowly, reducing the range of conditions under which civilization can function.

A crowned woodnymph hummingbird, whose iridescent feathers and rapid metabolism exemplify the fine-scale specialization that drives tropical biodiversity.
If coccolithophores were to collapse at scale, oceanic biomass would decline sharply with them. Some models suggest losses upwards of 30% of fish, marine mammals, and seabirds. One billion humans rely on fish as a primary protein source. Yet this is not a simple chain of cause and effect. It is part of a broader, interconnected unraveling.
Coccolithophores, of which roughly four hundred species have been described, with many more likely undiscovered, offer a way to understand biodiversity. They are only one example, but they reveal the kind of invisible labor I’m trying to learn how to see. It is the network of relationships that allows living systems to function, adjust, and recover over time.
As we begin to descend the volcano, this idea becomes easier to grasp without definition. Biodiversity reveals itself through motion and interaction. In ants and beetles, fungi and soil, birds and rivers, estuaries and mangroves, the same dynamic plays out again and again. Life sustaining life, quietly and collectively, across scales we rarely notice.
There is a discipline that tries to grapple with this truth directly. Ecological economics is the academic study that treats economies as embedded within nature and dependent on living systems. Ecological economics actually IS economics, because the very definition of economics is the study of unlimited wants in a world of limited resources. Ecological economics just accounts for irreversible failures in the system that provides humans with almost all their wealth. Even here, however, the limits are obvious. No model can integrate the full web of interactions that make ecosystems work. We can gesture toward value, but we cannot total it. The relationships are too many, too dynamic, too alive.
Biology and ecology are not blind to this complexity. They have revealed extraordinary detail in peer-reviewed work about food webs, nutrient cycles and feedback loops. Yet even with their collective depth of knowledge, we are nowhere near a unified understanding of how the biosphere functions as a whole, let alone how it fails. The systems that sustain life operate across scales of time, space, and interaction that exceed our ability to observe or integrate. Collapse is not a single mechanism or moment. It is a cascade, unfolding through relationships we only partially understand.
Imagine the biosphere as a sort of watch. A marvel of precision, assembled from hundreds of tiny gears, springs, and levers, each depending on the others to keep time. Now imagine that instead of hundreds of parts, there are billions. Imagine that many of them are alive. Some are brass, some liquid, some organic, all microscopic. They expand, contract, migrate, reproduce, and die. There is no instruction manual. No master diagram. And no watchmaker standing outside the system, immune to its failure.
If we could insert ourselves into that watch, though, we could make some sense of its systems. We might even learn that were we to tinker too much with it, we might bust the whole thing.
As we descend through the ecosystems of Costa Rica, that’s exactly what we’re beginning to do. We are beginning to glimpse that great clockwork; the gears that make a habitable world possible, a system we barely understand, and one we’re already altering faster than we can describe.
This journey begins on a volcano, but it will only end when I’ve reached that riverbank I cannot see or imagine this high above.
And yes — it turns out to be a hell of a lot of fun.
The Fungus Beetle and the Fungus
At night, the rain comes down hard at Rancho Naturalista—so hard that the four other guests cancel their plans for a guided night walk. Teylor, their guide, ends up with nothing scheduled and eggs me on to join him.
I grab my umbrella and headlamp and find him by the narrow trail leading into the trees. The sound of the rain is deafening. It doesn't quite drown out a Mottled Owl hooting in the distance.
While walking through the forest, Teylor's headlamp catches a beetle on a giant tree stump hanging over the slope.
It is already dead, fixed in place by the fungus that has overtaken it.
"It's ironic," Teylor says, lifting his flashlight toward the beetle. "It's a fungus beetle. They specialize in eating fungus. But sometimes it's the fungus that wins. They've killed the beetle, and now they're eating him."
The beetle is Cypherotylus vicinus. As a larva, this little guy lived buried inside rotting logs or hidden in the fungus-loaded leaf litter on the forest floor. Just chewing slowly, day after day, completely hidden away in its own private fungal universe.
The grown-up fungus beetles don't change their diet—they're still total fungus addicts, still mycophagous like before. The difference is that now you actually see them. They wander out onto the forest floor and move across dead wood and leaves. They eat whatever fungus is exposed—slime molds, small mushrooms, thin coatings on logs. They don't search so much as bump into food as they go.
Adult Cypherotylus vicinus have bright, striking coloration—red and orange, black and white. Here is an important rule of the forest: if you live on the ground and are colorful, you're probably poisonous. But if you're colorful and slow, you're probably deadly.

Pleasing fungus beetle (Cypherotylus vicinus) killed by cordyceps fungus on rotting wood.
Vicinus, like other pleasing fungus beetles, are absurdly slow.
Fungus-eating animals are often chemically interesting. In the case of vicinus, whatever makes them deadly likely comes from what they eat: processed fungal compounds weaponized into first-rate defense.
As we study the crime scene more closely, we can see that the fungus itself is like a miniature forest of white stalks with pink heads. The center of this stalk forest appears to be the beetle. The fungal growth has burst through the seams of the hardened body and now spreads outward across the scene of the crime.
What happened here?
The vicinus beetle was probably just doing what he always does, trudging along at the pace of an elderly sloth, stopping here and there to munch some fungi. Somewhere along the way, he must have brushed up against spores of a cordyceps fungus.
You might recognize the word cordyceps from The Last of Us, the HBO series starring Bella Ramsey and Pedro Pascal, where a fictionalized cordyceps infects humans, hijacks their brains, wipes out individual will, and turns people into zombies whose only role is to spread the fungus.
The human part is fantasy. The basic mechanism is not. Cordyceps infect the beetle because the beetle moves, and it infects beetles like vicinus because they spend their entire lives among fungi: ready-made transport, the way a burr catches in mammal fur.
After a few minutes of studying the crime scene, Teylor moves on, leaving the stump behind and continuing down the trail.

Moving effortlessly between surface and soil, the Costa Rican Red Leg Tarantula (Megaphobema mesomelas) operates both aboveground and within the hidden world below.
Beetles represent the most expansive exploration of animal life on Earth. About 400,000 species have been described, but coleopterists believe there are over two million beetle species in the world. They account for a whopping 40% of all known insects and 25% of all described animals on Earth. These insects occupy nearly every place where there is organic matter. They thrive in forest canopies and on desert sands, in river margins and rotting logs, deep soil and inside animal burrows. They basically live in almost every land and freshwater habitat. If coccolithophores represent a small number of species that have global reach on the biosphere, beetles are the opposite. They have global impact because of their diversity, their dazzling specialization.
The tiniest beetle in the world is a featherwing beetle. It's so small, at just 325 micrometers, you can just barely see it with the naked eye. I once saw a pair of titan beetles, almost seven inches long, not including legs. But I was absolutely horrified to learn that when I approached them, they could fly. The clanking sound of their stiff mechanical wings is the soundtrack to my own personal hell.
Some beetles live their entire lives inhabiting just one plant species. Others show up just when something is at a precise stage of decay, like when wood softens. They disappear as soon as it collapses into soil. Their body plans follow their specialized functions. Some are shaped with flattened forms to slide beneath bark, others are heavily armored to push through soil. Some are elongated to navigate tight crevices, and then, of course, there are those oceanic-like streamlined swimmers that patrol the margins of lakes and marshes.
Together, these millions of beetle species form a living architecture.
They aren't interchangeable. Most of them only do one thing, and often only briefly. Dead material gets reduced. Nutrients get moved. Energy goes up the food chain. It happens everywhere, constantly, without any coordination.
A few weeks ago, I was walking the sand dunes of Death Valley after dark when I noticed something tiny lumbering across the sand.
I lay down to take a closer look, hoping to send a photo to Jane and Kellan to compete with their cat pics. It was a pea-sized creature covered in long white hairs, making it look like a mythical mammal in miniature. It was Edrotes ventricotus, the Hairy Robot Darkling Beetle.
This species comes out at night and slowly munches the scattered desert debris into soil. They are so specialized, they live only among the loose sand of the Mojave and Sonoran deserts.
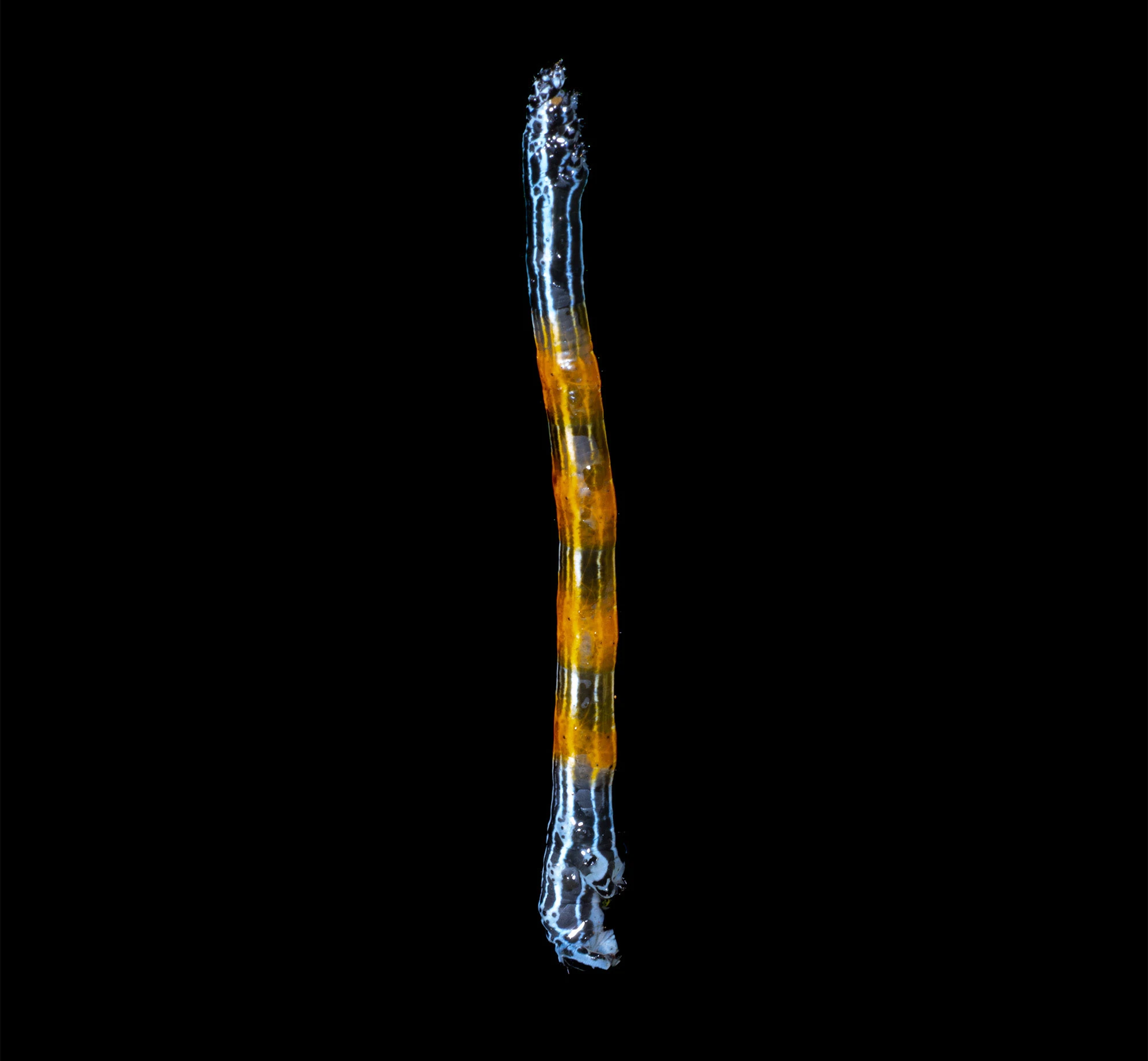
Hanging by a single strand of silk on a rainy night at Rancho Naturalista, this inchworm-like caterpillar turned out to be Simena luctifera, a Neotropical erebid moth in the family Erebidae.
Biologists will often say that beetles are the most important organisms on Earth. What makes them so critical is how their huge numbers explode into hyper-specialized roles, and how that specialization keeps ecosystems stable and the carbon cycle operating correctly on land. Without beetles, carbon gets stuck in frozen ground, locked in wood, pooled in waste, or dumped out fast when forests crash. Beetle diversity keeps those failures from happening all at once. Different species show up at different moments: one for a particular fungus, another for a certain stage of decay, others for dung, seeds, carrion, or soil. The constant movement of carbon in the natural world is essential for life; keeping too much of it out of the atmosphere is critical.
But beetles do more than stabilize climate. They keep soil working, stop forests from filling up with debris, hold pest outbreaks in check, and make sure nutrients make it back to plants. Without them, entire ecosystems would fail.
Among those ecosystems is our global agricultural output. Our crops are assisted by fertile soils renewed by beetles. In fact, over 95% of all crops depend on healthy soil, amended by beetles and other insects, fungi, and microbes. Dung beetles are considered essential in pasture productivity; they are among the primary controllers of livestock parasites like flies and nematodes, and they are essential in returning animal dung back into the soil.
Beetles even act as pollinators to some of our crops, including fruit trees, spices, palms, and magnolias. Entire forests persist because beetles keep many of the systems from falling completely out of control.
The biosphere, and the basic underpinnings of human civilization—from food security to clean air and water, as well as climate regulation itself—rely disproportionately on these six-legged hidden workers.
Farther up the trail, Teylor hoots something at me, which I can barely hear. I'm fussing with my headlamp, which flickers and goes dim.
As I turn on my phone's flashlight as a backup, my umbrella catches the dirt wall beside the path, scraping a thin layer of wet soil.
In the blue light of the phone light, I can see pale threads running through the dirt. The same ones I remember seeing while digging for worms as a kid in Minnesota.
The same filaments that fed the beetle.
The same network that eventually consumed it.
The same fungal network upon which all life depends.
Alien Face in the Rain
I am fussing with my reading glasses, which are beading with raindrops and fog. I can barely see Teylor's headlamp in the distance.
"Erik! Come here!"
I follow the trail to Teylor, who has stepped uphill into a rocky streambed that intersects the path.

Ridge-headed salamander (Bolitoglossa colonnea) balanced on a glossy leaf in the rain.
He is ecstatic with his find. A tiny salamander sits on a broad leaf about a foot above the riverbed. It is completely still. Bronze and blue fleck its pinkish body. Its tiny padded feet meld into the leaf's surface, and it wears a bumpy crown on his forehead.
We had just been staring at fungus bursting from a beetle's body. Now we are staring at a creature that lives because of that same underground architecture.
It's a Ridge-headed Salamander, Bolitoglossa colonnea. Only the second salamander Teylor has ever seen — and the first time either of us has encountered this species. Years ago in the Amazon I found a close relative, Bolitoglossa palmata, and I know this one instantly by the strange geometry of his face. The mouth is wide and slightly skewed, the head flattened and angular, as if shaped to clasp twigs in the dark. When I lean closer, rain dripping off my glasses, he looks uncannily like Nien Nunb, the Star Wars Sullustan who co-piloted the Millennium Falcon during the Battle of Endor.
There is something older in that face, too. Not that we descend from salamanders — we don't — but both of us reach back to the same early experiment in walking. Four-limbed vertebrates pulling themselves from tidal flats, bodies low and slick, learning gravity for the first time.
The fact that this is only Teylor's second salamander says more about Costa Rica than about him. In the Pacific Northwest, if I flip logs in spring or kneel beside a creek, I can usually find one: a flash of orange beneath bark, a coiled body under a stone.
Here, it's different. All forty-four Costa Rican species are lungless. Nearly all live in the trees. They spend their lives in the cloud forest canopy, moving through bromeliads and wet branches far above eye level. To see them regularly you would need ropes, harnesses, and a tent strung a hundred feet up in the mist.
Even there in the heights, they breathe through their skin. They depend on moisture. They remain creatures of water.

Phytolacca rugosa blossoms.
What unites all land-dwelling salamanders is that they inhabit substrate. They live in matter in transition: soil becoming forest, leaf becoming humus, wood becoming fungus. They dwell where life is being taken apart and reassembled.
And beneath that transition lies the meshwork that makes their world possible — the one I had just witnessed a few moments ago.
The relentless rain drives water into the soil. A centimeter below this drenched surface, delicate branching filaments of fungus — hyphae — extend from their tips, advancing cell by cell. They lengthen. They branch. They split and rejoin. Each tip softens what lies ahead and bends toward moisture and faint traces of food. They emit tiny electrical flickers that travel through the threads, signals bouncing back and forth through jungle and grassland. Within each filament, liquid flows steadily, sending nutrients through the same underground network.
When compatible filaments encounter one another, they pause. Each carries its own genetic signature — a quiet declaration of who it can join with. Some fungal species possess not two sexes, but hundreds or even thousands of mating types. When two suitable threads meet, they recognize one another chemically. Their walls soften. They fuse.
Underground, threads find one another.
They touch and merge, opening at the seam until their interiors run together. What began as two continues as one, extending outward through the soil.
It is quiet choreography in the dark.
Rain drums on my umbrella while, beneath my boots, thousands of microscopic courtships unfold.
Romance in the mud.
What appears to be many threads is often one organism.
They move through soil the way rivers move through landscape, following moisture, bending around stone, slipping into hollow twigs. Every tip presses forward into what can be dissolved, borrowed, remade.
Below the forest's spectacle of trunks and leaves, a quieter architecture spreads, filament by filament, contact by contact, stitching substrate into something living and shared.
A single gram of forest soil can contain meters of hyphae. Multiply that across an acre and the network becomes continental. Globally, fungal biomass is immense — comparable in scale to the mass of animals. While we count big animals like deer, jaguars, cattle, and ourselves as the visible engineers of landscapes, fungi are building planetary infrastructure in the dark.
Can you imagine explaining this in an airport lounge?
"Yes, the invisible threads beneath the forest weigh more than all the world's animals."
Security would escort me out.
The largest known mycelium individual, a Honey Fungus, exists back home in Oregon. It stretches three and a half miles wide beneath Malheur National Forest and may be 8,000 years old. One organism. Miles across. Older than most human civilizations.

Violet Sabrewing resting between flights, a flash of iridescent violet suspended in the green understory.
These threads lace through root tips, wrapping them, entering them, braiding themselves into living tissue. Tree roots don't stop at their own tips. They braid themselves into fungal threads, trading contact for reach, turning a modest root system into something vastly larger than the plant could manage on its own.
The fungi do the wandering. They slip into tight mineral cracks and through softening wood, pulling in phosphorus, nitrogen, zinc, copper — elements trees cannot easily pry loose for themselves. A tree can photosynthesize all day and still come up short on those raw materials. So it pays. Sugars made from sunlight move downward, out of leaves and into soil, feeding the mycelium that forages on its behalf. What looks like separate trunks aboveground is, below the surface, more like an exchange floor — carbon moving one direction, minerals the other, each side dependent on the other's labor.
The network carries warnings. When a leaf is chewed by insects, chemical shifts ripple down into the roots and outward through the fungal web. Neighboring plants adjust before the damage spreads. Leaves toughen. Defenses rise. The message travels underground first.
The network does more than pass along warnings. It coordinates growth. It redistributes stress. It shapes who thrives and who merely survives.
In grasslands, fungal threads link roots across entire meadows, helping plants share water during drought. In forests, they help decide which seedlings establish and which fail. Some trees funnel carbon generously. Others conserve. The network shifts in response. Sugars move toward the shaded, the stressed, the newly germinated. Nutrients flow along gradients of need.
A forest canopy rises from decisions made belowground. What we see in trunks and crowns is the outcome of exchanges carried out in darkness.
Hyphae sense changes in moisture, chemistry, and damage. During dry spells, water moves laterally through shared connections. When one plant is attacked, chemical signals spread through the web before leaves show visible harm. The network distributes resources, buffers stress, and absorbs shock. The forest stands because of decisions made between fungal filaments and the trees and plants themselves.
Carbon moves through these connections as well. Older trees send excess sugars toward shaded seedlings. In dense forests where young trees might otherwise starve, this quiet redistribution shapes survival.
Different species share the same networks. Fir may link to birch, pine to shrub, grasses to wildflowers. What looks separate aboveground becomes, belowground, a living conversation conducted in carbon and mineral.
This infrastructure stabilizes soils, regulates nutrient cycles, governs energy flow, and stores carbon. Beneath every field of wheat, every orchard, every city reservoir, the same architecture operates.

Golden Tortoise Beetle in the rain.
Just moments ago, we were looking at a dead beetle, a dismantler of wood. Its mandibles break cellulose into smaller pieces. It does not finish the work alone.
When a tree falls, beetles carve their galleries first. Then the mycelium enters the grain. Wood pales and softens. Structure loosens into soil. Carbon chains break. Nutrients free. Forests regrow after fire and hurricane because fungi reduce what came before into accessible form.
The planet's nutrient economy runs on fungal metabolism. What we call soil is, in large part, the proud work of mycelium.
For more than 400 million years, fungal filaments have been transacting with plant roots. Before forests, there were fungal threads. Before soil, fungal enzymes softened rock into sand. Animals built ecologies atop substrates fungi had already shaped.
When humans began planting grain, we stepped into an ancient partnership. Most of the crops that feed the world — wheat, rice, corn, beans, vegetables — still rely on fungal threads to gather nutrients their roots cannot reach alone. Strip away the fungi, and harvests grow fragile.
Nearly every calorie that has sustained agricultural civilization has passed, somewhere along its chain of dependency, through fungal networks.
Civilization rests on harvests.
Harvests rest on soil.
Soil rests, in no small part, on mycelium.
Each year plants push enormous amounts of carbon belowground. Not all of it lingers, but fungal networks help trap a portion of that flow, weaving it into soil in forms that do not immediately return to air. When those networks are broken — by plowing, by clearing land, by drenching fields in chemicals — the chemistry shifts. Soil that once held carbon begins releasing it. What should function as a reservoir becomes a leak. The damp earth beneath my boots is not scenery. It is part of the planet's climate machinery.
History has already shown what happens when soil fails. Irrigated fields in ancient Mesopotamia slowly salinized and lost productivity. In the Maya lowlands, forest clearing and erosion turned into decline. When the living fabric beneath a civilization thins, yields falter first. Then stability follows.
We have cataloged roughly 150,000 fungal species. Many researchers suspect the real number may be in the millions. Each one occupies a narrow role, tuned to a particular forest, grassland, or wetland. Each influences how nutrients circulate, how soil aggregates, how ecosystems absorb shock. Reduce that diversity and the system does not collapse instantly. It tightens. Its range of response narrows.
So what does that tightening mean for the biosphere, and for us?
Water that once infiltrated begins to run off, dragging soil with it. Farmers compensate with more fertilizer to coax the same yield from thinner ground. Streams carry heavier sediment loads. Reservoirs silt faster. Forests endure drought with less resilience because roots no longer tap into shared reserves.
Carbon that once settled into soil returns more quickly to the atmosphere. Nutrients move faster and leak away. Systems that once buffered disturbance begin to loosen.
At first, the shifts resemble inconvenience: declining yields, stressed trees, higher input costs. Over decades, they accumulate into instability. Erosion advances. Margins narrow. Capacity diminishes.
Fungal diversity is already thinning. Industrial tillage severs hyphal networks. Fungicides suppress symbiotic partners alongside pathogens. Land conversion fragments underground continuity. Climate shifts alter the moisture regimes fungi depend upon. A complex system is already growing simpler.
Already, about ten percent of all land on Earth is used for agriculture, and another thirty percent for grazing and pasturelands. Another two percent is covered in concrete urban zones. And even of the thirty percent that remains forested, much is logged — not truly wild. An enormous percentage of fungal networks are already fragmented, damaged, burdened, or lost.
Agriculture grows more brittle. Crops rely increasingly on synthetic nitrogen and phosphorus, inputs derived from fossil fuels and mined reserves that are finite, fundamentally dangerous to our climate, and geopolitically uneven. As natural nutrient exchange declines, fertilizer dependence rises. Costs increase. Runoff intensifies. Downstream dead zones expand.
Water systems destabilize. Soils with reduced fungal biomass absorb less precipitation. Floods sharpen. Droughts deepen. Reservoirs accumulate sediment more quickly.
Carbon cycles accelerate. Fungal networks move carbon into stable pools of soil. When those pathways thin, carbon lingers less securely underground and returns more readily to the atmosphere. In that way, the health of mycelium networks is one of our defense systems against runaway climate change.
When these networks decline, the effects accumulate gradually. Yields decline. Insurance rates rise. Rural economies tighten. Migration pressures build where soil productivity falters. Food insecurity expands outward from vulnerable regions. Last summer, on the islands of Malta and Gozo — where thin soils and water scarcity make agriculture precarious — this same pattern kept revealing itself.
A civilization built atop simplified soil systems must work harder and spend more to replace processes that once operated quietly beneath its feet. Each replacement carries cost. Each cost compounds.
The question becomes: how much ecological redundancy can be stripped away before the capacity to absorb disturbance narrows beyond recovery? Mycelial networks are not thinning in isolation. Forests are fragmenting. Insects are declining. Soils are eroding. Hydrologic cycles are destabilizing. It is the convergence among all these things that is difficult to grasp. These systems interact across scales, microbial, botanical, atmospheric, in ways that exceed our current capacity to fully model them. Even the atmospheric systems described in IPCC reports, vast as they are, represent only one layer of a far more entangled biospheric architecture.
The salamander lifts one forelimb and advances it across the leaf's midrib. Rain beads along its back and slides away.
No vertebrate group is unraveling as quickly as amphibians. Forty percent are now considered at risk of extinction. In the span of my own lifetime — since the years I spent digging in the dirt looking for worms — roughly two hundred species have vanished, some of them the most astonishing in their color and form.
One of the chief culprits is chytrid fungus. Humans have ferried it across continents, often without knowing it. The pathogen attacks the skin, the very surface amphibians use to breathe and regulate water. In some places the impact is shockingly swift: populations that once seemed secure crash in a matter of weeks.
Their decline registers disturbance moving through the system.
They are our siren.
Salamanders falter while forests still appear intact. Frogs vanish while watersheds continue to flow. Their collapse tells of the stress lines of the near future.
They carry early tremors of deeper instability, faint as the drip of water from the leaves above us.
The salamander stares back at us from his perch. Teylor crouches beside me, silent, his headlamp catching the rain. The creek continues its conversation with the rain in the dark.
Under this slope lies a living lattice of fungal diversity stabilizing soil, storing carbon, routing nutrients, sustaining forest resilience.
Civilization imagines foundations of stone and steel.
The real foundation hums beneath leaf litter in filament and exchange.
We turn back toward the trail. Beneath us, hyphae extend, fuse, divide, trade carbon, glue soil, and hold the slope that holds the river that feeds the valley that feeds the city.
The threads continue negotiating survival.
And ours.
Part II: Lago Angostura >
